LSR injection mold directly determines whether products can pass rigorous testing in the research and development of thick walled silicone components in the medical and high-end industrial fields.
If you are locked into creating thick walled silicone parts that can both pass the USP Class VI biocompatibility testing and withstand 200°C, you would be addressing three main issues:
Certified silicone is passed, but post-molding exudates are out of limits. Heat resistance test is passed, but dimensional deformation of a sample occurs after sterilization. Traditional manufacturing process cannot simultaneously satisfy both perform-ance requirements.
The above problems can not only cause project delays and cost overruns but even lead to the failure of the product launch.
Core Answer Summary
| Key Challenges | Core Technology Solutions | Expected Value |
| Biocompatibility vs. Heat Resistance Conflict | Mold runner optimization + precision temperature control system | Eliminates the risk of material thermal degradation and ensures no exudates. |
| Internal Defects in Thick Walled Parts | Vacuum-Assisted Exhaust + Staged Pressure Holding Process | Eliminates porosity, achieves 100% density, and meets pressure-resistant sealing requirements. |
| Dimensional Drift Due to Repeated Sterilization | Shrinkage pre-compensation design + low internal stress molding | Dimensional tolerance controlled within ±0.05mm after 121°C moist heat sterilization. |
| Multi-Cavity Quality Consistency | Hot runner balancing technology + online viscosity monitoring | Ensures CpK ≥ 1.33 during continuous production of multi-cavity molds. |
Key Takeaways
- Just choosing the right materials won't be enough:
Even if the materials used are certified biocompatible, this certification may be invalidated if the molding processes are not carried out correctly.
- Every aspect of the mold defines quality:
Features like a runner design without any dead angles and accurately treated surfaces contribute immensely to the heat resistance longevity of thick walled parts.
- A data-based approach for stable results:
The successful large-scale production of high quality, multi-cavity molds relies on exact thermal balancing of the molds and constant process monitoring.
- Working with experts is the fastest way through:
For the timely production of complex parts that comply with USP Class VI and heat resistance requirements, the joint design of materials, molds, and processes cannot be overemphasized.
Why Trust This Guide? JS Precision’ Experience In LSR Injection Mold Services
With more than 15 years of experience in precision injection molding, JS Precision is dedicated to addressing your LSR injection mold and medical grade silicone component R&D challenges.
We have produced more than 300,000 high precision custom parts and have served over 1,000 clients worldwide. Thanks to our 99.2% on-time delivery rate, you will be able to avoid project delays and keep the projects moving efficiently.
In addition, we are certified with ISO 9001:2015 and ISO 13485:2016 medical quality systems.
Besides that, our injection mold tooling design and manufacturing operations meet worldwide medical manufacturing standards and are completely traceable, so you can be sure that compliance risks in the mass production of medical components will be a thing of the past for you, and you will be able to pass certification with every batch without worrying about compliance issues.
We have helped medical device manufacturers in Europe and North America solve the problems of biocompatibility and heat resistance of thick walled LSR seals and thereby helped them increase the yield rates from 65% to 98% and reduce the mass production cycle times by 40%.
Our real-world case studies support you in cost-saving through reducing trial-and-error and achieving mass production profitability quickly.
We have shrinkage rates of more than 50 different LSR materials in our database, covering a wide range of hardnesss from 20-80 Shore A and thickness of walls from 1-10 mm.
All the process parameters are confirmed with DOE experiments helping you with accurate data to avoid the losses of reworks due to the deviations in the dimensions and controlling production costs.
Our engineering team is available 24/7 for performing an online DFM analysis and also they can answer in 15 minutes your technical questions.
We accompany you through all the stages from designing an injection mold until the delivery of the mass production. This way we help you to overcome your quality concerns related to medical grade components and at the same time, you can effortlessly and easily move forward with your projects.
Download the LSR injection mold medical application white paper now and get free personalized basic process consultation and material selection advice.
Can Custom Silicone Parts Pass USP Class Vi And 200°C Sealing?
Sure, the secret of whether custom silicone parts can meet these two extremely strict standards at the same time is in the combination of material formulation and molding process.
Would you like us to assist you with the process verification step and the injection mold tooling design? After that, the full-process compliance testing according to ISO 10993-1 standards is done to ensure product certification success.
Synergistic Effect of Material Formulation and Process
Actually, a lot of suppliers are not able to offer both heat resistance and USP Class VI certification at the same time, which may lead to project delay.
We conduct DOE experiments to set the vulcanization parameters exactly. In this way, your custom silicone parts will be able to maintain 70% of tensile strength even after 7 days of heat aging at 200C and passing biological evaluation, which significantly lowers the risk of failing certification.
Sealing Performance Verification
After the molding stage, all custom silicone parts are subjected to helium leakage testing (leakage rate 1 10 mbarL/s). This ensures their sealing will be dependable even at 200°C. The LSR injection mold services operating under full process control exclude the possibility of seal failure.
The Trade-off Between Platinum Vulcanization and Peroxide Vulcanization
Since platinum vulcanization of LSR generates no byproduct precipitation, it is a better option for your USP Class VI scenario, peroxide vulcanization may be a better choice if you want more heat resistance but it needs a secondary vulcanization step.
We will give you a formulation proposal and match medical grade mold designs to your product requirements, then select the best solution.
How Do Precision Injection Molds Survive Repeated Sterilization?
Dimensional changes caused by multiple sterilizations may cause product assembly failures.
Precision injection molds accurately predict the LSR shrinkage rate (2.0% -3.5% after 121 ° C wet heat sterilization), optimize the process to reduce internal stress, and ensure product size stability and pass rate compliance after sterilization.
Prediction of Internal Stress and Shrinkage
Through simulation of internal stresses with high precision mold flow software the residual stresses are controlled below 5MPa.
Furthermore, the combination of residual stress relief with precision injection molds dimensional compensation results in a critical dimensional change of 0.02mm even after 50 sterilizations cycles, fully eliminating dimensional drift.
Annealing Process: Post-treatment to Eliminate Internal Stress
Aiming at the production of high precision parts, we integrate a 150°C, 2-hour annealing process, which releases stress resulting from molding orientation.
This action greatly enhances the product's dimensional stability after sterilization and guarantees that it can meet medical grade precision standards leading to successful acceptance testing.
Material and Process Matching
We support your precision injection mold design with our nearby shrinkage rate knowledge base of more than 50 LSR materials. LSR injection molding services are also capable of simultaneously adjusting to the best process, thus reducing the times for trial molding and dramatically shortening the development cycle.
![]()
Figure 1: A technical diagram of the Liquid Silicone Rubber (LSR) injection molding process, showing A/B material cylinders, a static mixer, and the injection into a mold.
How Do LSR Injection Mold Services Ensure Both Biocompatibility And Long-Term Thermal Stability?
Biocompatibility and lasting heat resistance are the main features of medical grade products.
LSR injection mold operations employ platinum-catalyzed high purity materials that, along with a tightly controlled temperature, can guarantee the complete cross-linking of the material and the absence of precipitation of low-molecular-weight substances in order to comply with the regulations.
Purity Ensured from Materials to Finished Product
Our LSR silicon base materials are of USP Class VI standard, so the only source of contamination is the design injection molds stage, which we have eliminated.
The mixing of the materials takes place in a Class 10,000 clean room. Our LSR injection mold services keep the purity control level very high at all times to make sure that there are no issues with the biocompatibility certificatio.
Process Parameter: Vulcanization Window Control with Precision
Vulcanization curve analysis is our instrument for determining crosslinking parameters. By controlling injection molding temperature fluctuations within 2°C, This will result in 100% complete vulcanization of the material and the absence of any low-molecular-weight leaching.
LSR Injection Mold Process Parameters and Performance Correspondence
| Process Parameters | Vulcanization Temperature (°C) | Vulcanization Time (s) | Mold Temperature Fluctuation (°C) | Shrinkage Rate (%) | Exudate Grade |
| Conventional Process | 190±5 | 60 | ±5 | 3.5-4.0 | Grade 3 |
| CNC Process | 185±2 | 90 | ±2 | 2.0-2.5 | Grade 1 |
| Thick-Wall Dedicated | 180±2 | 120 | ±3 | 2.2-2.8 | Grade 1 |
| Multi-Cavity Mass Production | 185±2 | 90 | ±2 | 2.0-2.5 | Grade 1 |
| Medical Grade Standard | 160-200 | 60-150 | ±3 | 2.0-3.5 | ≤ Grade 1 |
Submit your component specifications and receive free customized process solutions and cost estimates for LSR injection mold services.
What Do LSR Injection Mold Services Include For Medical-Grade Heat Resistance?
LSR injection molding services comprise to the following threemain modules: selection of heat resistant materials, designing a thermal balanced mold of thick walls, and controlling the processes in closed loop. T
hese offer end-to-end support for the mass production of medical grade heat resistant components, thereby solving the problem of balancing dual performance needs.
Technical Depth: The Molding Process is a Guarantee of Performance
Even if you pick 250°C-resistant materials, injection temperature drifts or mold contamination may still cause the loss of certification. Our LSR injection mold services keep the temperature deviations small (within 2°C) and along with medical grade mold design, they minimize risk initial points.
Material Validation: Heat Aging DataSupport
Before mass production, all LSR materials are subject to 200°C, 168-hour heat aging test in order to ensure the retention of tensile strength and elongation at break. LSR injection mold services deliver full material validation for your custom silicone parts, preventing risks from lack of heat resistance.
Maintaining No-variation of Vulcanization in Thick Walled Parts
For parts having thicker walls than 5mm, zoned temperature control is independently used to keep the difference in mold cavity temperature 3°C.
As a result, the very thick walled core does not get incompletely cross linked. On top of multi-cavity LSR mold production services, stable mass production is attained and efficiency is increased.
How To Design Injection Molds Without Biocompatibility Risks?
When you are designing injection molding dies, it is a must to use a full thermal runner system and be sure that there are no dead zones.
Here at our company, we can assist you in removing melt stagnation zones, stopping LSR high temperature degradation, eliminating toxic byproduct migration, ensuring product biocompatibility, and preventing the failure of certification.
Zero Stasis Runner Geometry Design
By means of mold flow analysis, we adjust runner parameters for optimum melt flow and uniform shear heat at 200°C, eliminating any potential material degradation by a stagnation time of over 30 s since the runner design of injection molds is directly related to determining the biosafety baseline of your product.
Cold Runner vs Hot Runner Selection Strategy
In the case of products with very high biocompatibility requirements, go for a fully hot runner system so as to get rid of the contamination caused by recycled runner heads.
If opting for a cold runner system, we strictly limit the recycling rate to 15% and, besides medical grade molds, the use of design injection molds to ensure the product compliance.
Mold Material: Compatibility Verification with LSR
We decide on S136 or 420SS corrosion resistant mold steel that is CNC machining and vacuum heat treated to stop metal ion migration from high temperature mold precipitates, thus not affecting your product's biocompatibility testing. Besides using precision injection molds, we also use design injection molds to ensure mold safety.

Figure 2: A metal multi-cavity mold is shown with two precision-made transparent plastic components placed on it, demonstrating the outcome of the molding process.
Why Do Medical Grade Molds Require Special Surface Treatments For Heat-Resistant Applications?
Medical grade molds surface treatment plays an essential role in demolding and quality of your product. For instance, if the surface roughness Ra > 0.1m, the products are highly susceptible to demolding damage.
To remedy this, we provide ultra fine polishing and nano coating treatments to not only prevent demolding damage but also to enhance product yield.
Surface Roughness Ra 0.05m - Why is it so Important?
To raise the surface hardness of medical grade molds to HV 2000 or even beyond, we resort to PVD or diamond like carbon coatings.
After 3000 high temperature cycles, the surface still has low friction, which can help you achieve non destructive demolding. Besides that, it ensures the product appearance and mass production stability.
Choosing a Coating: DLC vs CrN
Based on product characteristics, our coating selection aligns to your customized solutions for medical grade molds in conjunction with LSR injection molding services.
Demolding Angle: Geometric Compensation for Less Adhesion
Basically, if you are dealing with deep cavity or undercut products, we elevate the demolding angle of medical grade molds to 1.5°-2°, and redesign the ejector pin layout to make demolding less resistant. At the same time, the structure of injection molds is being optimized to significantly lessen product demolding damage.
Medical Grade Molds Surface Treatment Parameter Table
| Surface Treatment Type | Roughness Ra (μm) | Surface Hardness (HV) | Heat Cycles | Coefficient of Friction | Applicable Scenarios |
| Conventional Polishing | 0.2 | 800 | 1000 | 0.3 | General Industrial Parts |
| Precision Polishing | 0.1 | 1000 | 2000 | 0.2 | High End Industrial Parts |
| DLC Coating | 0.05 | 2000 | 3000 | 0.08 | Medical High Viscosity LSR |
| CrN Coating | 0.05 | 1800 | 3500 | 0.12 | Fluorosilicone Wear Resistant Parts |
| Medical Grade Standard | ≤0.05 | ≥1500 | ≥3000 | ≤0.1 | Medical USP Class VI |
Get a free quote for medical grade molds surface treatment solutions, clarifying mold processing costs, lead times, and quality standards.
How Do Multi-Cavity LSR Mold Production Services Keep Quality Consistent?
Multi-cavity LSR mold production services employ separate hot runners and a mold temperature balancing system to restrict temperature variations between cavities to within 2°C, thus avoiding under curing and over curing and ensuring uniform product performance across all cavities and compliance with mass production requirements.
Stable CpK 1. 33 across Cavities
When employing 32-cavity/64-cavity multi cavity molds for mass production, we analyze the pressure and heat distribution of each cavity simultaneously.
This way, the hardness variability within a batch is ensured to be 2 Shore A, which is a medical grade standard. Multi cavity LSR mold production services are capable of stable mass production.
Hot Runner Balancing
By using independent temperature controlled valve type hot runners, we maintain the difference in injection pressure and filling rate between cavities within 3%, thereby preventing dimensional deviations.
Multi-cavity LSR mold production services, along with newly designed injection molds, are able to achieve runner balancing.
SPC Combined with Full Inspection
Throughout production, measurements and hardness testing are done on a random basis every two hours. At the same time, CpK changes are tracked by SPC.
The very important parts are subjected to 100% full inspection by using optical sorting machines. Multi cavity LSR mold production services when combined with LSR injection mold services result in comprehensive quality control and waste reduction.
Case Study Of JS Precision: Breakthrough In High Heat Resistance Of Thick Walled LSR Seals
Client Background
A European medical equipment manufacturer once faced the challenge of developing thick walled LSR seals for cardiac assist devices, which required USP Class VI certification and over 2000 high-pressure sterilization cycles at 134 ° C to maintain sealing performance.
However, due to insufficient technology from the supplier in the early stages, the project was stalled and cost overruns occurred, which may also be a dilemma you may face.
Challenges Encountered
The top three issues that customer had in the beginning are:
Micropores in the 6mm thick wall that led to the seal bursting during sterilization, range of inner diameter shrinkage 0.2mm after sterilization which resulted in assembly failure, and very high molding temperature resulted in low molecular-weight precipitation with biocompatibility tests failure.
Solutions
1.Venting Design:
We employ design injection molds technology to create a layered vacuum venting system so the thick walled area can be evacuated to -0. 1MPa during mold closing, completely eliminating internal pores and preventing seal failure due to bursting during sterilization.
2.Dimensional Accuracy:
We use precision injection molds to accurately predict shrinkage rate, which combined with overmolding technology and database, reduces the inner diameter tolerance zone from 0. 2mm to 0.05mm, thus preventing dimensional drift after sterilization and effectively solving assembly failure problems.
3.Molding Process:
Our LSR injection mold services involve closed loop pressure control system to keep mold temperature at 185±2°C, thus material cross linking at the best point is locked. By this, you will be able to avoid precipitates resulting from over sulfurization and guarantee your product will be biocompatibility tested successfully.
4.Medical Standards:
Our injection mold tooling process is performed according to medical manufacturing standards and quality is comprehensively guaranteed, components are ensured to be of the highest quality and your product gets successfully to pass USP Class VI certification, thus warding off project delays caused by certification failures.
Final Results
Ultimately, we help our clients achieve three core benefits:
- Their components pass USP Class VI test at the first try with no cytotoxicity.
- We help their seal retention rate still be 95% after 2500 sterilization cycles at 134°C.
- Their yield rate is increased from 65% to 98%, so that they can start clinical trials 6 weeks earlier. These are the real values of our partnership that we provide you with a reliable model for the mass production of similar medical thick walled LSR components.
Core Challenges and Solutions Value Comparison Table
| Challenge Type | Pre-Solution Defect Rate | Solution | Post-Solution Defect Rate | Cost Reduction (%) | Cycle Time Reduction (%) |
| Biocompatibility Exudation | 35% | Flow Channel Optimization + Precision Temperature Control | 2% | 30 | 40 |
| Thick-Wall Porosity Defects | 40% | Vacuum Degassing + Staged Pressure Holding | 1% | 25 | 30 |
| Sterilization Dimensional Drift | 30% | Shrinkage Compensation + Low Stress Molding | 1% | 20 | 25 |
| Poor Multi-Cavity Consistency | 25% | Hot Runner Balancing + Online Monitoring | 2% | 15 | 20 |
Submit your sealing component drawings to receive a free customized mass production solution and DFM report for the same LSR injection mold.
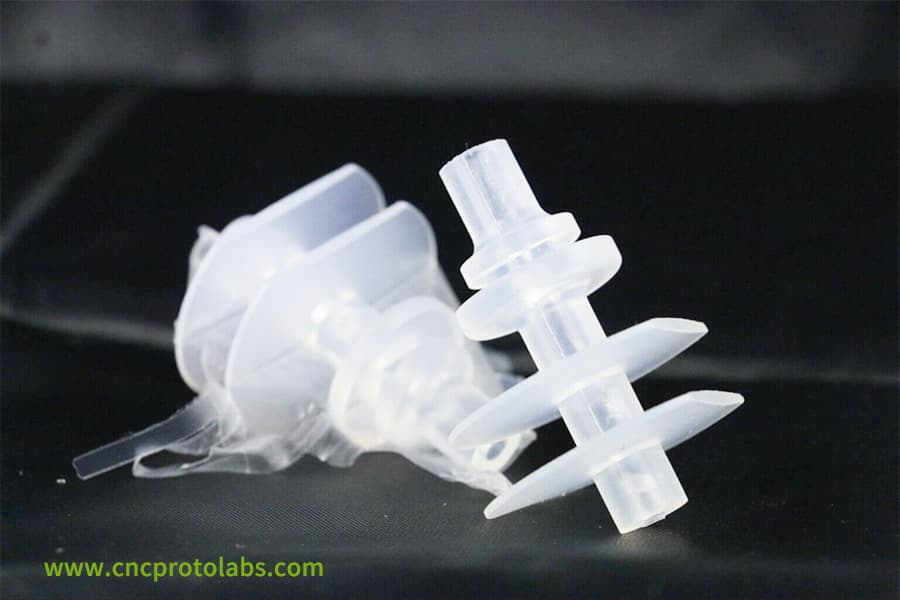
Figure 3: A close-up of a complex, spiral-shaped transparent plastic component, likely a mixer or impeller for a medical device.
FAQs
Q1: What is the minimum wall thickness you can achieve for LSR injection molding?
Generally, our LSR injection molds have a minimum wall thickness of 0.5mm. However, such thin sections require a very high precision mold and a vacuum-assisted system in order to completely fill the cavity with the material.
Q2: Can you process FDA-approved LSR materials?
We are capable of processing a variety of platinum-catalyzed LSR materials that are compliant with FDA 21 CFR 177.2600 and USP Class VI, thereby meeting medical and food contact standards.
Q3: How to avoid incomplete internal curing in thick walled LSR parts?
By conducting a staged pressure holding and extending vulcanization time as well as using temperature control of the mold, we ensure a uniform heat transfer which in turn avoids incomplete internal curing of thick walled LSR parts.
Q4: How to ensure consistent weight in each cavity of a multi-cavity mold?
By both an accurately balanced runner system design and online weight monitoring, we keep the injection amount deviation in each cavity within 0.5%, which results in weight consistency over a range of cavities.
Q5: What shrinkage rate is typically allowed when designing an LSR mold?
Usually, the shrinkage rate of LSR materials ranges from 2.0% to 3.5%, depending on factors like the type of material, the thickness of the walls, and the process. To get the accurate value, we will provide the mold flow analysis results.
Q6: How long does it take to manufacture a medical grade LSR mold?
Generally, it takes 4-6 weeks to produce standard medical grade molds, although this timeframe varies with the mold complexity and number of cavities. To accelerate your approval process, we'll send you a DFM report.
Q7: Will LSR material harden for 200°C high temperature applications?
High grade LSR material fully vulcanized is very stable and after aging at 200C its hardness changes minimally (5 shore A). If the material is not vulcanized properly, there will be a significant hardness increase.
Q8: Can you provide secondary overmolding services for LSR components?
We are able to offer secondary overmolding of LSR onto metals and rigid plastics. Also, proper design of the mating surfaces is carried out so that high bonding strength and sealing performance are ensured.
Summary
Under the dual tests of biocompatibility and heat resistance, only by deeply integrating the precision design of LSR injection mold, rigorous surface treatment, and closed-loop control of process parameters can truly qualified medical grade thick walled components be delivered.
This is not only a competition of equipment, but also a game of engineering experience.
For example, if you are looking for a mass production solution of the high requirement LSR components (thick walled, USP Class VI, repeated sterilization scenarios), you may get in touch with us for a DFM report.
Our engineering and technical team based in China will offer you full-process data support starting from mold design to mass production, enabling you to overcome technical and quality barriers, effectively lead project implementation, and reach profitability.
Disclaimer
The contents of this page are for informational purposes only.JS Precision Services,there are no representations or warranties, express or implied, as to the accuracy, completeness or validity of the information. It should not be inferred that a third-party supplier or manufacturer will provide performance parameters, geometric tolerances, specific design characteristics, material quality and type or workmanship through the JS Precision Network. It's the buyer's responsibility Require parts quotation Identify specific requirements for these sections.Please contact us for more information.
JS Precision Team
JS Precision is an industry-leading company, focus on custom manufacturing solutions. We have over 20 years of experience with over 5,000 customers, and we focus on high precisionCNC machining,Sheet metal manufacturing,3D printing,Injection molding,Metal stamping,and other one-stop manufacturing services.
Our factory is equipped with over 100 state-of-the-art 5-axis machining centers, ISO 9001:2015 certified. We provide fast, efficient and high-quality manufacturing solutions to customers in more than 150 countries around the world. Whether it is small volume production or large-scale customization, we can meet your needs with the fastest delivery within 24 hours. Choose JS Precision this means selection efficiency, quality and professionalism.
To learn more, visit our website:www.cncprotolabs.com




