Medical insert molding is a fundamental technology for creating accurate packaging in the manufacturing of medical components. In medical device manufacturing, if the insert is displaced or the bonding fails, it does not only result in a very high scrap rate (as high as 5%-8%), it may also risk the patient's safety.
So, how do you get micron-level packaging precision without harming the delicate electronic components or precision metal needles?
As one of leading medical device component manufacturers, JS Precision provides customers with high-yield, low-risk customized solutions through advanced insert molding technologies and a strict 13485 medical manufacturing system, effectively addressing their core pain points in medical component production.
Core Answer Overview
| Dimensions | Core Technology Solutions | Value Delivery |
| Precision Control | By the usage of low-pressure filling (keeping the pressure below 0.05N) along with sequential valve hot runner technology to totally remove melt impact. | Insert displacement risk cut down by 90%, ultra-precise packaging stability at the 0.01mm level guaranteed. |
| Quality Compliance | Carrying out IQ/OQ/PQ in a very strict manner under the ISO 13485 environment and risk management based on ISO 14971. | Achieve 100% batch consistency and deliver high-standard, fully traceable lifecycles (DHR) that are compliant with regulatory requirements. |
| Cost Optimization | Deploy 6-axis robotic auto loading and hot runner DFM design to minimize the wastage of costly medical-grade resins. | Reduction of injection molding cycle time by about 25%, a major step in optimizing unit processing costs and production efficiency. |
Key Takeaways:
- Ultimate Precision:
The displacement of the smallest inserts is controlled with a very high precision within a tolerance range of 0.01mm through the use of pressure compensation and precision limiting technologies.
- Compliance Assurance:
ISO 13485 certification not only eases legal compliance risk but also ensures that the production process is in line with major medical market access standards worldwide such as FDA/CE.
- Cost Reduction and Efficiency Improvement:
The combination of automated integration with hot runner systems not only raises the production capability but also decreases the loss of high-priced medical-grade resins such as PEEK by 15%-20%.
- Technological In-Depth:
Expert packaging service for even the most complicated parts such as CGM sensors. E.g. high hermeticity and strong bonding of the metal-plastic interface.
Why Choose JS Precision's Medical Insert Molding Service?
Medical component makers selecting a trustworthy medical insert molding service must consider the fact that this impacts product safety, compliance, and competitiveness in the market.
JS Precision has turned into the favorite partner of leading medical device manufacturers worldwide because of its industry knowledge and capabilities.
We run our operations in line with the requirements of FDA 21 CFR Part 820 medical device manufacturing standard, thereby providing you with a wide range of risk-reduction and benefit-generation opportunities from technology compliance cost, and delivery points of view.
The years of insert molding technology research and development at JS Precision have resulted in low-pressure filling and sequential valve hot runners, which are very accurate medical component production assisting technologies.
These technologies aid in performing the displacement control of tiny inserts inside very narrow tolerances as low as 0.01mm, thus completely solving the usual industry problems of insert displacement and adhesion failure.
On top of that, the fact that it has an extremely high CpK index (greater than 1.33) and has shown no variation in performance in repeatability testing for three consecutive batches (at least 1000 pieces per batch) effectively makes the assurance of product consistency and quality fluctuation risk reduction.
Real-life examples confirm even more the importance of our partnership. A client was producing a minimally invasive surgical guidewire assembly, whose metal needle diameter was only 0.3mm. The traditional injection molding led to a displacement of 0.12mm, causing a 7% scrap rate.
On the other hand, JS Precision's personalized medical insert molding solution resulted in a displacement of the insert down to 0.01mm, and the client was able to reach a stable yield of 99.2%.
This not only saves the environment but also enables the company to be more productive and at the same time keeps the product within the required standard, which is basically giving you the real-life benefits of the tech advantages.
If you are struggling with precision and compliance issues in medical component packaging, contact a JS Precision engineer for free case details to intuitively understand how we can solve your pain points and create value.
Why Choose Services Under The ISO 13485 Medical Manufacturing System?
The essence of manufacturing medical parts is ensuring compliance and safety. A 13485 medical manufacturing system offers this fundamental assurance. It is not just a document, rather, a quality that is embedded in the whole production chain. Insert molding, being a very important stage, has to be run under this system.
JS Precision is fully committed to the ISO 13485 requirements framework for IQ/OQ/PQ validation and the ISO 14971 approach for risk management. By means of process control, we determine injection molding failure modes, thus helping to ensure that performance/ tolerances/ remain unchanged over the course of multiple batches.
This way we support client's efforts to obtain FDA/CE global market access approvals.
Detailed Validation Process (IQ/OQ/PQ) for Consistency Assurance
IQ, OQ, and PQ three stage validation is the "legal passport" for medical devices entering the European and American markets, confirming equipment compliance, stable operation, and performance standards.
My CpK index > 1. 33, with three consecutive batches (1000 pieces per batch) achieving repeatability testing compliance, effectively guaranteeing product consistency.
In a nutshell, this stands for a daily meal production that requires pre-production hygiene surveying and ensuring taste consistency in every batch during production so that the product is available on the market and consumers can feel safe.
Production Risk Prevention Management Based on ISO 14971
We identify potential hazards in five areas: people machines materials, methods, and environment. We use the FMEA assessment method, along with a Class 10,000 cleanroom (particle count < 3520 particles/m³, 0.5μm), to eliminate quality risks at the source and minimize the chances of product recalls for our clients.

Figure 1: A spacious, clean manufacturing workshop with orderly rows of large equipment, operated by personnel in protective attire, indicating a high-standard production environment.
How To Solve The Displacement Of Inserts In Precision Components Through Medical Insert Molding?
Displacement of inserts is one of the biggest issues in medical insert molding. The delicate parts such as long needles and electrodes in micro precision components may be displaced by the impact of molten material leading to product safety issues.
JS Precision has come out with a dedicated solution that can be a good compromise between the accuracy and speed of insert molding technologies.
We install a filling system with low pressure and slow speed and also a hot runner sequence valve system to make sure the force on the insert is less than 0.05N, thus fundamentally eliminating the risk of displacement. It is the result of two main technologies:
Pressure Compensation Technology and Sequence Valve Control
We employ pressure schemes broken down into segments (each change < 0.5MPa), combined with a valve needle opening delay time of 0.01s-0.05s.
With the help of multi point injection self centering, we neutralize the side force of the melt on one side, thus preventing the insert moving out of place. This is the main benefit of our insert molding technologies.
In other words, it is similar to gently pouring water into a very narrow container. By changing the speed and angle of water flow, you can prevent it from splashing and the container from shaking ensuring a very precise and leak free pouring.
Precision Positioning of Micro Inserts and Mold Fine tuning
Our positioning pins are manufactured with tolerance control of +0.002mm/-0. Besides that, the hardness of the mold insert can be as high as 58-62 on the HRC scale. Grinds down to the micron level provide a zero wobbling locking of the insert.
Together with professional injection molding inserts technology, this results in a great improvement of encapsulation accuracy.
| Technical Parameters | Control Standards | Traditional Process Level | Customer Benefits | Applicable Scenarios |
| Installation Force | ≤0.05N | 0.1-0.2N | Reduces insert deformation and displacement | Microelectrode, metal needle encapsulation |
| Valve Needle Opening Delay | 0.01s-0.05s | Over 0.1s | Balances melt impact force | Ultra-thin wall component molding |
| Positioning Pin Tolerance | +0.002mm/-0 | +0.01mm/-0 | Zero-wobbling positioning | Precision sensor encapsulation |
| Mold Insert Hardness | HRC 58-62 | HRC 50-55 | Extends mold life, ensures stable precision | Mass production projects |
If you are facing the challenge of precision component insert displacement, submit your product drawings, and JS Precision will customize a dedicated medical insert molding solution for you and provide a free cost calculation.
Which Insert Molding Technologies Are Suitable For Packaging CGM Continuous Glucose Monitoring Sensors?
Complex components such as CGM sensor housings often involve the integration of multiple materials including metal, ceramic, and electronic components, requiring extremely high compatibility with insert molding technologies.
Ability of JS Precision, one of leading medical device component manufacturers, to fulfill these requirements hard with their flexible solutions.
Protecting Heat Sensitive Components with film Assisted Molding Technology
The film assisted molding process uses polyimide (PI) films to shield sensor parts from thermal shock. PI layers are kept between 25m and 50m thick. This helps maintain cavity temperatures at 15°C to 20°C.
And this setup reduces heat exposure for delicate circuits and coatings. It seems the effect is mostly noticeable in high pressure environments. And the thin film acts as a barrier, keeping sensitive parts from direct contact with extreme heat.
Application of precision Limiting Inserts in miniature Sensor Packaging
Space in miniature sensors is tight, so electrode compression matters. We keep the ratio below 5% and above 3%, limiting post packaging impedance shifts to under 0.5%.
By using mechanical inserts, we prevent compression during molding from affecting electrical readings. Hard to ignore how this directly supports CGM accuracy. There's no room for variation in alignment or pressure.
How Can Professional Medical Insert Molding Service Solve The Problem Of Adhesion Between Metal And Plastic?
Insufficient bonding between metal and plastic can easily cause medical parts to crack and peel, which is why it is important to follow safety regulations. JS Precision uses its expertise medical insert molding service, among other techniques, helps the bonding strength and also meet the high standards of injection molding insert quality.
Bond Strength Improvement by Using Insert Preheating System
We use a preheating system for the insert so that the insert temperature can be maintained at 100°C ± 5°C. The plastic will cool down normally and condense without any disturbance from the insert. Polymer molecular chains will have an easier time entering the metal micropores this way.
As a result, the amount of thermal stress is decreased and an interfacial bonding strength of 15 MPa or more is obtained.
Surface Modification Technology Effect on Hermetic Sealing
We use a plasma treatment to increase the metal surface tension above 48 dynes/cm. It is a way of promoting chemical bonding and ensuring an hermetic sealing of IP67 or higher level. The sealing comes with a helium leak detection rate of < 1 10 mbarl/s and it is resistant to multiple autoclave sterilizations.
| Processing Method | Core Parameters | Bonding Strength | Airtightness Standard | Sterilization Tolerance Cycles | Applicable Materials |
| Insert Preheating | 100°C ± 5°C | >15MPa | IP65 | 50+ cycles | Stainless Steel, Medical Plastics |
| Plasma Treatment | Surface Tension >48 dynes/cm | >20MPa | IP67 | 100+ cycles | Titanium Alloy, PEEK, PC/ABS |
| Untreated | Room Temperature | <5MPa | IP54 | Less than 10 cycles | General Metals, Plastics |
| Plasma + Preheating | 100°C ± 5°C, Surface Tension >48 dynes/cm | >25MPa | IP68 | 200+ cycles | Various Medical Metals, High Performance Plastics |
Want to know how to improve the metal plastic bonding strength of your product through medical insert molding service? Contact a JS Precision engineer for one-on-one professional consultation.
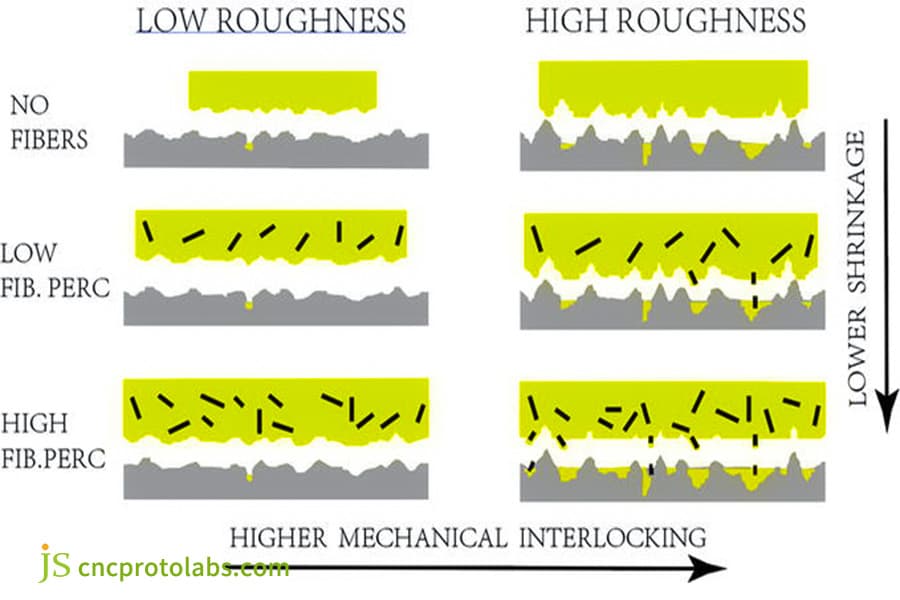
Figure 2: A technical diagram illustrating how fiber content and surface roughness influence the shrinkage and mechanical interlocking (adhesion) of injection-molded parts.
Which Insert Molding Technologies Can Address The Integration Challenges Of Complex Medical Components?
Surgical instrument handles, sensor housings, and other complex medical components often involve multi material integration, requiring extremely high stability from insert molding technologies. JS Precision being one of leading medical device component manufacturers, offers a range of answers to these integration issues via flexible solutions.
Comparison of Vertical and Horizontal Injection Molding Solutions
We provide vertical and horizontal injection molding equipment, which have clamping forces from 30T to 250T. Vertical machines rely on gravity for self centering of long needles and multiple inserts, with insert gravity offset 0.001mm. Horizontal machines are a better match for large housing encapsulation, balancing efficiency and precision.
6 Axis Robotic Automated Loading and Improved Yield
We present 6 axis robotic automated loading, which leads to a repeatability accuracy of 0.02mm and a pick-and-place cycle that is 40% faster than manual loading. Also, non contact loading results in less damage to the delicate medical surfaces and thus leads to a higher yield of medical device injection molding.
Multi Material Integration (Metal/Ceramic/Electronic Components) Packaging Strategy
We change the parameters of the process to significantly raise interfacial shear strength by 30%-50%, exactly adjust the CTE difference of dissimilar materials, avoid cracking, and accomplish the one cycle fusion of metals, ceramics, and medical grade plastics, which can increase production efficiency.
How Can Excellent Medical Device Component Manufacturers Optimize DFM To Reduce Costs?
Optimizing Design for Manufacturability (DFM) is the basis of cost cutting and efficiency improvement. JS Precision as a professional medical device component manufacturer, assists its clients in controlling medical device injection molding costs through minute changes that still do not compromise the product performance.
Insertion Standardization Recommendations and Mold Complexity Reduction
We share with our clients insert designs suggestions, e.g. keeping chamfer angle between 0.5° and 1° and maintaining the insertion depth to diameter ratio close to the ideal 2:1. These changes lead to a shorter mold processing time and injection molding simpler insert processing.
The Value of Hot Runner Technology in Medical Resins
Medical resins like PEEK are very costly. Besides that, traditional methods lead to waste of as much as 30%. Our hot runner technology cuts down waste rate to less than 3%, which means a quick recovery of initial mold investment and lower unit costs for long term mass production.
Cycle Time Optimization Solution
By using conformal cooling and high thermal conductivity materials, we are able to have a Reynolds number (Re) of above 4000 for cooling water channels, which means the injection molding cycle time is reduced by 10-15 seconds. This allows more production per hour, saving costs and improving efficiency.
How To Evaluate The Long-Term Delivery Capability Of Medical Insert Molding Suppliers?
Mass production of medical components necessitates the stable supply capability of the factory. JS Precision is tightly following the ISO 13485:2016 standard and has wonderful scalability even from the prototype to the mass production, thus satisfying the production needs of the customers on a long term basis.
Scalability from Single Cavity Prototype to High Efficiency Multi Cavity Molds
We are equipped to handle 1x4, 1x8, and 1x16 multi cavity mold, with dimensional deviations between cavities <±0.01mm.
An extremely well equipped internal tooling room facilitates quick mold maintenance and iteration, thereby meeting different requirements from prototype to mass production.
Digital DHR Recording and Full Production Cycle Traceability
We realize digital DHR recording through an MES system, where the parameter acquisition frequency is 10ms and the traceability records are archived for over a decade. Each part is traceable back to the raw materials, operators, and process parameters, thus being in line with the regulatory and FDA audit requirements.
Want to assess our long term delivery capabilities? View JS Precision's multi cavity mold mass production case studies and contact us for detailed delivery capability specifications and certifications.

Figure 3: A close-up view of an operator’s hands working on an injection molding machine that is producing cylindrical medical components.
JS Precision Case Study: High Precision Packaging Of 0.3mm Guidewire Insert
Miniature insert packaging is a major issue for customers. Conventional medical insert molding often leads to the dislodging and distortion of metal inserts which have a diameter of less than 0.5mm. JS Precision is able to address this issue completely by using the most advanced insert molding technologies.
Challenges Faced
The client was making a minimally invasive surgical guidewire assembly that consisted of metal needles with a diameter of only 0.3mm. Injection molding in the usual way caused a displacement of 0.12mm which was beyond the 0.05mm tolerance, the result was a 7% scrap rate.
In addition, it was quite challenging to get the ISO 13485 certification, which is a requirement for market access in Europe and the United States, and which also causes waste and delay in delivery.
Solution
JS Precision gathered a professional engineering group after the client's needs were understood in order to design a one-of-a-kind medical insert molding solution that is perfectly compatible with the client's products.
- Our low pressure, slow fill method reduces peak pressure by 40%, completely removing the problem of melt impact and preventing the crazy displacement of metal pins caused by excessive force.
- We have created a precision limiting slider insert in conjunction with micron level grinding technology to control the tolerance of the locating pin within +0.002mm/-0, thus ensuring no metal pin movement inside the mold cavity.
- Hot runner sequential valve control technology enables us to open four valve pins after a certain delay (up to 0.01s), which helps the melts to converge at the same time from both sides of the insert and balance the lateral forces well.
- We ensure that the health safety requirements are met by following the ISO 13485 medical manufacturing system very strictly while implementing the three stage validation of IQ/OQ/PQ. As a result of FMEA, risk management has identified human, machine, material, method, and environment as the main risks of production, and steps have been taken against them to ensure continuous batches.
- At the same time, 6 axis robotic automatic loading technology helps to remove contamination risks and deviations of position caused by manual intervention as well as increasing production efficiency.
Final Results
Displacement was lessened to 0.01mm, an increase in yield up to 99.2%, monthly scrap losses that went down by $2 000 the product not only got ISO 13485 certification and 10,000 fatigue tests but also made it to the European and American markets, and the injection molding cycle got shortened by 25%.
Are your products also facing the challenge of micro insert encapsulation? Submit your detailed requirements, and JS Precision will customize a dedicated medical insert molding solution for you, providing free DFM analysis.

Figure 4: A transparent tube with a blue connector and a thin metallic wire, exemplifying a high-precision minimally invasive surgical guide wire component.
FAQs
Q1: What is ISO 13485 certification in medical insert molding?
ISO 13485 is a quality management standard designed specifically for the medical device industry. It defines the medical insert molding requirements along with other manufacturing processes to ensure that everything is compliant and safe. This certification also helps medical devices gain access to global markets.
Q2: How does automated loading help with quality?
The 6 axis robot has a positioning accuracy of 0.02mm, which removes human error and contamination, improving the uniformity of medical insert molding products as well as enhancing production efficiency.
Q3: How to ensure the airtightness of the metal plastic interface?
Plasma treatment (surface energy48 dynes) together with insert preheating are used to activate chemical bonding of the surfaces. As a result, the interfacial adhesion will have the quality of IP67 waterproof, and can also endure several sterilization cycles.
Q4: How to optimize medical injection molding costs through DFM?
Among all the ways of performing design for manufacturing (DFM), selecting the optimal wall thickness, minimizing the number of undercuts, standardizing the specifications of the injection molding inserts, and using hot runner molds can help reduce medical injection molding costs by 15%-25% and increase production capacity.
Q5: How does JS Precision ensure product traceability?
Using the MES system, a digital DHR record is created, through which any batch of parts can be traced back to raw materials, operators, and process parameters, thereby complying with ISO 13485 standards.
Q6: How can we tackle the difficulties in molding high performance plastics like PEEK?
Maintaining the crystallinity of PEEK according to standard is key to reducing molding defects. This can be achieved by using 200°C high temperature mold temperature controller and refining insert molding technology parameters.
Q7: Are you capable of dealing with micro packages requiring 0.1mm precision?
Indeed, our CGM sensor packaging is an example of micron level packaging where through precision positioning and low pressure filling technology we manage to achieve 0.1mm level accuracies as well as protection of the core components.
Q8: How long does it usually take to go from prototyping a new project to mass production?
Generally, it takes 15-20 days for prototype mold making. After the IQ/OQ/PQ verification, mass production expansion will take 45-60 days. The timeline can be changed based on the requirements for ensuring rapid delivery.
Summary
The most important factor in medical component packaging by precision is picking an expert medical insert molding service with whom you can have a reliable partnership.
JS Precision is still customer oriented, using state-of-the-art insert molding technologies, and a strict 13485 medical manufacturing system to target pain points like insert movements and compliance, offering economical one stop solutions.
From prototyping to mass production, we are able to precisely adjust to support you in conquering the market. Get in touch with JS Precision engineer right away for a free DFM analysis report.
Disclaimer
The contents of this page are for informational purposes only.JS Precision Services,there are no representations or warranties, express or implied, as to the accuracy, completeness or validity of the information. It should not be inferred that a third-party supplier or manufacturer will provide performance parameters, geometric tolerances, specific design characteristics, material quality and type or workmanship through the JS Precision Network. It's the buyer's responsibility Require parts quotation Identify specific requirements for these sections.Please contact us for more information.
JS Precision Team
JS Precision is an industry-leading company, focus on custom manufacturing solutions. We have over 20 years of experience with over 5,000 customers, and we focus on high precisionCNC machining,Sheet metal manufacturing,3D printing,Injection molding,Metal stamping,and other one-stop manufacturing services.
Our factory is equipped with over 100 state-of-the-art 5-axis machining centers, ISO 9001:2015 certified. We provide fast, efficient and high-quality manufacturing solutions to customers in more than 150 countries around the world. Whether it is small volume production or large-scale customization, we can meet your needs with the fastest delivery within 24 hours. Choose JS Precision this means selection efficiency, quality and professionalism.
To learn more, visit our website:www.cncprotolabs.com





