Medical injection molding plays a big role in turning the medical device idea into a real product ready for production.
For example, Your Class III medical devices gave the desired results in animal trials, but the injection molding supplier's failure to provide complete batch traceability records and an ISO 10993 biocompatibility report, caused a delay of six months in your FDA 510(k) submission.
This problem doesn't happen very rarely, rather it is one of the most common issues that medical device developers face when they are trying to turn sophisticated designs into compliant products.
Ordinary injection molding services cannot comply to the very strict regulatory requirements for risk management and documentation completeness, only professional medical injection molding can meet these requirements.
Core Answer Summary
|
Core Question
|
Key Answer
|
|---|---|
|
ISO 13485 Medical Injection Molding Definition
|
A dedicated process for medical devices with end-to-end quality control and traceability.
|
|
Core Material Requirements
|
Must be ISO 10993 or USP Class VI certified.
|
|
Key Precision Indicators
|
Cpk ≥ 1.33, micron level tolerance control capability.
|
|
Main Cost Components
|
Mold costs account for the majority, unit cost decreases as production volume increases.
|
|
How to mitigate risks
|
Choose suppliers with early intervention and a scientific injection molding validation system.
|
Key Takeaways:
- Compliance comes foremost:
Suppliers not having ISO 13485 certification are unable to fulfill FDA or MDR requirements for traceability and change control. This is essentially why many customers' FDA applications get blocked.
- Materials can make or break the situation:
Certified biocompatible materials are the only way. PEEK and medical grade silicone being the most popular choices, directly impact the safety of the product and its access to the market.
- Cpk is what determines accuracy:
Fully electric injection molding machines are instrumental in reaching micron level tolerances and sustaining a process capability index (Cpk 1.33). In simple terms, this is equivalent to 99.993% of the products conforming to the specifications.
- Early steps in making changes cut costs:
Modifying the mold design at the DFM stage can result in 15% - 30% less costs of the mold, thereby customers can lessen their initial investment.
Insights From JS Precision’s Experience In Medical Injection Molding: Why Is It Trustworthy?
Engaging in the medical device sector to the extent of pushing your products to large scale production is a scenario where a medical injection molding partner is precisely what you need. Ideally, such a partner should be capable of significantly lowering your approval risks and smoothly carrying out your projects.
JS Precision has a long and deep experience in supplying compliant injection molding solutions for the medical device industry to more than 500 clients, out of which over 30 projects involved Class III implantable devices.
We are capable of guiding you towards successful FDA 510(k) and EU MDR approvals. Our success is very much hinged on our in-depth knowledge of industry's pain points and expert handling of the overall process.
ISO 13485:2016 mandates that medical injection molding needs the institution of a complete quality system in the whole process. It is also our main philosophy, which we follow strictly in our production so that you can stay compliant.
Perhaps you could identify with the above situation:
A client focusing on cardiovascular devices went through an FDA approval delay of four months for the Class II catheter product because of the original supplier's failure to provide complete batch traceability records which directly resulted in the client missing a very valuable market window.
When such situations arise, JS Precision could be a reliable partner for you. The client who contacted us was at the same time the one whose supply chain we quickly mapped out to be a full traceability system wherein we captured every operation not only in raw material warehousing but also all the way to finished product delivery.
Achieving the goal however required going beyond just record-keeping - we also provided a biocompatibility report from ISO 10993 in support of your submission. Ready in only a couple of months, your approval became a reality which is now in your hands to make up for lost time and capture market opportunities.
Opting for JS Precision is not simply about getting injection molding services that comply with standards. On the contrary, it entails thinking ahead, discovering potential risks and lessening the overall expenses.
We even get involved during your product design phase to help you optimize and so that you don't have to pay the high costs of mold modifications after the fact, as well as not making unnecessary investments.
The results are evident: working with us can raise your FDA approval rate by 92% so that you don't have to waste time and energy on approval delays.
Cut total costs by 20% on average so that you stay in control of your budget while your product competitiveness is enhanced.And reduce delivery time by 30%, thus allowing you to get your products to market faster than your competitors.
If you are also struggling with compliance, precision, or cost issues related to medical injection molding, you can download our "ISO 13485 Medical Injection Molding Compliance White Paper" to intuitively understand how we help similar clients overcome FDA approval challenges and reduce injection molding costs.
What Is ISO 13485 Certified Medical Injection Molding And How Does It Differ From Standard Injection Molding?
ISO 13485 certified injection molding medica is a highly regulated operation meant only for medical devices.
The main distinction between a normal injection molding process and one compliant with ISO 13485 is the implementation of a comprehensive quality traceability system along the whole chain, rather than just focusing on the dimensions and visual aspects of the product.
This is the factor that leads directly to a product being able to successfully pass FDA and MDR approvals.
Full Chain Control and Traceability System
Complete chain controllability is the very heart of ISO 13485 certification.
Raw material warehousing and the production process to finished product delivery, every step is documented and a complete batch history record (BHR) is formed which enables not only rapid traceability of the problem source but also reduction of batch risks.
Simply put, it is like making a full "born-to-factory" record for each batch of products. When a problem occurs, this method ensures that the source of the problem is localized with great accuracy, like how a package's delivery logistics are tracked.
Core Difference Comparison: Compliance vs. Appearance
|
Comparison Dimensions
|
General injection molding
|
ISO 13485 medical injection molding
|
|---|---|---|
|
Main Issues
|
Dimensional tolerances, shrinkage, flash, and other appearance plus basic dimensional issues
|
Compliance, full chain traceability, risk management, and complete documentation
|
|
Quality Assurance Methods
|
Just simple finished product sampling inspection, no systematic control
|
Complete control of all processes, ISO 14971 risk management standards implemented
|
|
Traceability
|
No batch traceability, unable to identify the origin of the problem
|
100% batch traceability, establishing a complete BHR file
|
|
Regulatory Clearance
|
Fail to meet FDA, MDR, and other medical regulatory standards
|
Fully meet the requirements for FDA 510(k), MDR, and other approvals
|
Who needs these services?
Medical device developers targeting FDA 510(k) or PMA submissions and planning to enter the EU MDR market should select medical injection molding services that are ISO 13485 certified, regardless of the type of medical product they manufacture. Compliance is a must for market access.
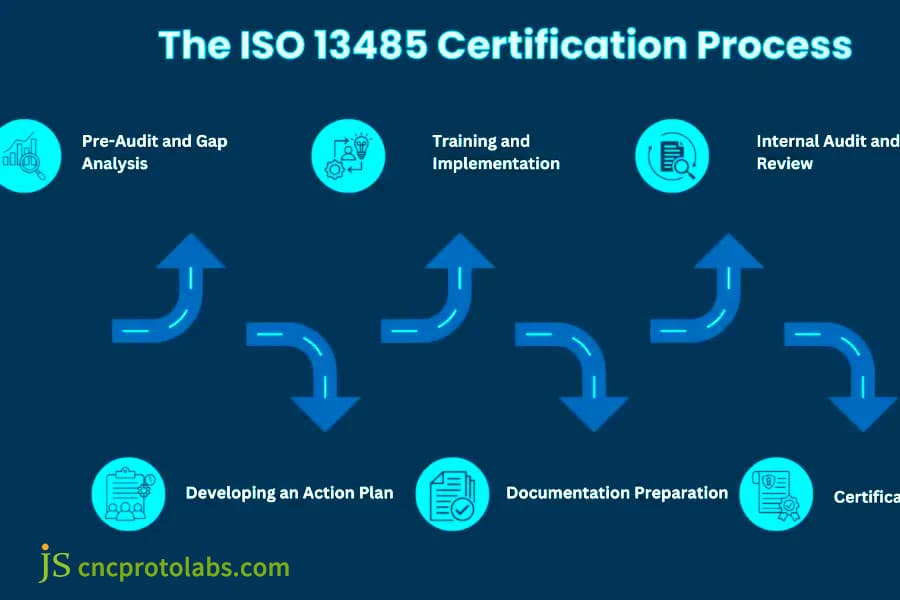
Figure 1: A flowchart detailing the ISO 13485 certification process steps, from Pre-Audit to Certification, essential for quality management in medical device manufacturing.
How To Select The Right Materials For Biocompatible Injection Molding For Medical Devices?
Materials are the major factor that directly influence the safety and suitability of injection molding for medical devices. It is necessary to order the use of polymers that are certified by ISO 10993 or USP Class VI, and also fulfill the FDA 21 CFR Part 820 requirements for medical materials.
Main Standards: ISO 10993 and USP Class VI Certification
Materials the suppliers should be asked to submit test reports and make sure they are in line with ISO 10993-1 covering cytotoxicity, sensitization, and systemic toxicity. USP Class VI certification is required for those devices that will be in contact with human living fluids, thus providing a higher level of safety.
Comparison of Properties of Four Commonly Used High Performance Medical Polymers
|
Material Name
|
Melting Temperature
|
Core Properties
|
Applicable Scenarios
|
Certification Status
|
|---|---|---|---|---|
|
PEEK
|
343°C
|
High temperature resistance, high strength, sterilization resistance
|
Implantable devices, orthopedic components
|
ISO 10993, USP Class VI
|
|
PPSU
|
280°C
|
Resistant to repeated steam sterilization, good toughness
|
Surgical instrument handles, medical containers
|
ISO 10993, USP Class VI
|
|
UHMWPE
|
130°C
|
Excellent abrasion resistance, low coefficient of friction
|
Orthopedic joint components, sliding mechanisms
|
ISO 10993
|
|
Medical Grade Silicone
|
150-200°C
|
Soft, good biocompatibility, excellent sealing
|
Seals, catheters, soft contact components
|
ISO 10993, USP Class VI
|
|
Medical Grade PC
|
260°C
|
High transparency, impact resistance, dimensional stability
|
Transparent instruments, diagnostic equipment components
|
ISO 10993, USP Class VI
|
Technical Challenges: Flowability Control of High Melting-Point Materials
High temperature materials like PEEK have very viscous melt during processing, which can easily cause defects such as insufficient filling and bold weld lines.
Thus, it is important to select a medical injection molding company that has high speed, high pressure, fully electric injection molding machines and screws that are specially designed.
Send us your device requirements and material preferences for free advice on selecting biocompatible material, which will help you avoid the usual mistakes in medical injection molding material selection and ensure that materials comply with ISO 10993 certification, which is the basis for subsequent compliance approvals.
What Technical Factors Define High-Precision Medical Device Injection Molding?
At the heart of the high precision injection molding for medical devices process lies the goal of reaching micrometer level tolerances and continuously producing in a stable manner.
A full electric injection molding machine represents the main machinery in the whole process, and its precision and steadiness significantly supersede that of conventional hydraulic presses.
It can cater to the stringent requirements of minimally invasive surgical instruments and implant grade parts and this is indeed one major difference between medical device injection molding companies that are professional and regular manufacturers.
Micron Level Tolerance Control: Hydraulic vs. All Electric
Medical grade products usually require micron level tolerances. The equipment precision difference between the two would directly affect the product quality:
- Hydraulic press: the tolerance limit is approximately 25m, it can be largely affected by the oil temperature, and the precision can fluctuate considerably, so it is only suitable for general medical consumables.
- All electric press: can reliably achieve tolerances of ±5μm to ±10μm, the position control accuracy can be as low as 0.001mm, it is not influenced by the oil temperature, and it can be used for high precision demands.
Design for Manufacturing (DFM) Optimization
In order to prevent wastage of the cost at the later stages of the product development, the design phase needs to be making changes to the injection molding process.
- Control uniform wall thickness within 1.5-2.5mm range to prevent shrinkage and warping.
- Allow chamfer radius to be 0.5mm so the part gets less internal stresses and injuries.
- Set draft angle to 0.5°-1° to get an excellent finishing surface.
Cpk Process Capability Index as a Main Performance Indicator
For medical grade manufacturing, a minimum Cpk of the critical dimension of 1.33 is targeted (which means 99.993% of the manufactured pieces meet the specification). JS Precision is always able to produce a critical dimension Cpk above 1.33 and sometimes it even reaches an even higher level of 1.67.
Another way to illustrate this is a factory producing parts with only 0.7 defective parts out of 10,000 parts, which is far beyond the pass/fail criterion of regular production, just like a top level sports player who maintains an exceptionally high shooting percentage.

Figure 2: A collection of transparent and precisely molded plastic components, demonstrating the high-quality output required for medical device applications.
How Can Medical Injection Molding Companies Solve Common Quality Defects Like Flash And Sinks?
Flash and shrinkage are two of the main defects of medical injection molding that not only affect appearance but also the safety of the product. Top manufacturers manage to minimize defects at the source by scientific injection molding, real-time monitoring, and early fault prediction.
Cavity Pressure Monitoring Technology
Shrinkage defects are mainly due to insufficient holding pressure or pressure changes. By putting pressure sensors in the cavity to measure the maximum pressure and keeping the pressure variation within 0.5%, one can drastically reduce shrinkage and prevent the occurrence of defective products.
Moldflow Analysis for Defect Prediction
Before the mold is opened JS Precision uses Autodesk Moldflow analysis to locate air traps and weld lines, thereby optimizing venting channels (0.02-0.03mm) and weld line temperatures to prevent defects and reduce debugging costs.
Three-Stage Validation System: IQ/OQ/PQ
Only through the implementation of this method can specialized medical injection molding companies guarantee stable production:
- IQ (Installation Qualification): Checks if the equipment has been installed according to the requirements and ready for use.
- OQ (Operation Qualification): Finds the process limit windows and defines the controllable ranges.
- PQ (Performance Qualification): Tests three successive production batches to verify batch quality.

Figure 3: A side-by-side comparison showing a complete blue plastic part and one with a “short shot” defect, where the mold cavity was not completely filled.
What Are The Challenges Of Multi Shot Medical Injection Molding For Complex Devices?
Multi component medical injection molding (two-color, overmolding) is a fundamental technology for the production of highly complex medical devices. The main problem is the interfacial bonding between two biocompatible materials to prevent delamination.
Multi component Technology: Application of Rigid Glue + Soft Glue
Overmolding technology is by far the simplest technique. Rigid substrates such as PPSU and PEEK are first injection molded, then soft materials like medical grade silicone and TPE are injected around to form parts like non-slip handles and sealing interfaces. This way, no further assembly is required, leading to higher efficiency.
Technical Solution: Ensuring Interfacial Bond Strength
JS Precision addresses the delamination issue by implementing two main measures:
- Plasma and corona treatments applied to the surface of the first injection raise the surface energy to more than 56 dyne/cm.
- Through mold flow analysis, the temperature is optimized so that the second injection material partially melts (0.1-0.3 mm) the layer of the first injection surface, thus forming a chemically stable bond.
Put simply, this is like "roughening" the contact surfaces of two materials and then "heating and bonding" them together, so that they become firmly stuck to each other. Just as sanding the surfaces to be glued makes the adhesive bond stronger and less likely to fail.
Cost Benefit Analysis: Reduced Assembly and Cross-Contamination
This technology is able to merge several components into a single one, thereby cutting down the number of assembly steps and labor costs. At the same time, it is able to decrease cross-contamination rates by more than 90% and raise production efficiency by 35%.

Figure 4: A detailed view of a complex metal injection mold with intricate channels and components, highlighting the tooling used in high-precision manufacturing.
How To Reduce The Total Cost Of Injection Molding For Medical Devices?
Mold expenses are 40%-60% of the entire cost of medical injection molding projects. Production volume drive down the cost per unit, getting a handle of scientific optimization techniques will measurably shrink your customer's investment.
Cost Structure and Economies of Scale Data
|
Annual Production
|
Mold Cost (Two Holes)
|
Unit Cost
|
Material Loss Rate
|
Total Production Cost
|
|---|---|---|---|---|
|
10,000 units
|
$15,000
|
$0.40
|
5%
|
$19,200
|
|
50,000 units
|
$15,000
|
$0.28
|
3%
|
$29,200
|
|
100,000 units
|
$15,000
|
$0.20
|
2%
|
$37,000
|
|
500,000 units
|
$20,000
|
$0.12
|
1.5%
|
$89,000
|
|
1,000,000 units
|
$25,000
|
$0.08
|
1%
|
$105,000
|
Hot Runner Economic Threshold
Cold runners have initially lower costs for production volumes below 20,000 units, hot runners, on the other hand, because they effectively cut down on material wastage, become more cost effective over the long term when production volumes go above 20,000-30,000 units.
Mold Structure Optimization Path
If possible, stick to the use of simple straight pull (two-plate mold) structures. Since structures involving features such as side core pulling and sliders will not only hike the mold costs by 15%-30%, but will also lengthen the production cycles, you should try to stay away from them unless they are really indispensable for your product.
Material Replacement and Process Adjustment Cost Reduction Case Study
JS Precision changed the raw material of the disposable surgical forceps from PPSU to a medical grade PC+ABS alloy and at the same time also optimized the cooling time (from 25 seconds to 18 seconds), thereby achieving the lowering of unit cost by 22%.
Provide your production volume, part specifications, and cost budget, and we will calculate the overall cost of injection molding for medical devices for you free of charge, helping you find the most suitable cost reduction solution and achieve a win-win situation for both cost and compliance.
Why Is Early Design Involvement Critical When Partnering With a Medical Injection Molding Medica Provider?
Getting the medical injection molding companies involved early in the design process can lower costs, increase producivity, and eliminate waste that arises from making changes at a later stage.
A lot of clients don't realize this resulting in their mold modification cost piling up and delivery being delayed. Certified companies for injection molding of medical devices would not only talk about it but also get involved in your design to save you from these risks.
Benefits of Design Optimization (DFM)
Our engineers at JS Precision get involved in the product design if necessary and optimize the structure of the product in the light of injection molding medical devices processes.
For instance, we combine several parts into one piece, optimize the locations of gates, decrease the cost of assemblies, and enhance the rate of production without defects.
Prototype Stage: Use of Rapid Prototyping
Thanks to clinical trials, mass production steel molds that cost a fortune will not be necessary. Our quick prototyping molds made of aluminum and mild steel can generate 50-500 samples within just 2-3 weeks, at 10%-20% of the cost of mass production steel molds, thus cutting the costs of trial and error greatly.
Supply Chain Transparency: Digital Traceability from Beginning to End
It allows for setting up of a full traceability system when you get involved from the very beginning. Through a link between raw material batch, process parameters, and quality inspection report using the MES system, a single click is enough to get data from the system which can be used for supporting FDA/MDR approval.
JS Precision Case Study: Reducing Comprehensive Costs By 23% For a Class III Implantable Peek Component?
Injection molding medical devices, especially for Class III implantable ones, must be done very carefully so that the devices comply with all the regulations, have high precision and don't cost too much. JS Precision understood their client's issues and were able to cut the client's costs with their targeted solution.
Challenges:
The customer whose orthopedic implantable devices provider encountered three main difficulties with their Class III spinal fusion device:
- A mold cost of $58,000 way over the figure on the budget.
- A unit price of $4.50 along with annual production of 20,000 pieces, leaving the product noncompetitive enough.
- As well as the previous supplier not being able to deliver ISO 10993 traceability documentation, which was a barrier for their FDA 510(k) submission.
Solution (JS Precision):
1. DFM Optimization:
The original side pulling slider structure was changed to a straight pull mold structure by simply rearranging the parting lines. As a result, the side pulling mechanism was completely removed.
This not only made the mold structure more straightforward but also increased its stability and lifespan. Through direct consequence, this led to a reduction in mold costs to $42,000 a 28% decrease, and helped the client in managing the upfront investment.
2. Material Assurance:
We obtained a PEEK material with ISO 10993 certification and provided the full batch certificates and biocompatibility test data.
Besides, we helped the client to finish the material validation to make sure that the product is in conformity with the FDA requirements for medical implant devices, which is the main reason behind the client's FDA 510(k) application.
3. Process Optimization:
Injection molding machine with the Fanuc all electric type was used. Keeping the mold temperature controller at 175C, it was possible to accurately control the parameters of material temperature, injection pressure, and holding pressure.
Apart from boosting the weld line strength from the design 45MPa to 62MPa thereby ensuring product strength for implantation requirements.
Cpk value of critical dimensions was also improved from 0.85 to 1.45, which is well above the medical grade production requirement of 1.33, hence a significant improvement in product yield.
Final Results:
The unit cost was hammered down to just $3.45 - that's a whopping 23% drop, which in turn helped save $21,000 speaking in terms of the annual cost.
The mold cycle was shortened to 8 weeks, FDA approval was received on the very first attempt, and the project was smoothly moving ahead.
If you also have medical injection molding needs for Class III implantable devices, submit your 3D drawings and product technical requirements to receive free DFM optimization advice and a preliminary quote. Directly replicate our cost reduction results and quickly advance your project.
FAQs
Q1: What is the core value of ISO 13485 certification?
It is very important to have traceability and risk control during the entire medical injection molding process chain for medical devices to be successful in the European and American markets. This also helps to get FDA and MDR approvals.
Q2: How to verify the biocompatibility of injection molded parts?
You should ask your supplier to give you ISO 10993 test report which contains cytotoxicity and sensitization tests and ultimately make sure material safety by carrying out random sampling of finished products.
Q3: What Cpk level is required for medical grade production?
Cpk 1.33 (99.993% product compliance) is necessary for medical grade devices and Cpk 1.67 is mandatory for implantable devices.
Q4: What are the typical mold costs?
A two-cavity medical mold normally costs around $15,000 to $50,000, implantable molds that are complex are more expensive.
Q5: What are the common defects in multi component injection molding?
The biggest problem is delamination and peeling which results from lack of adhesion between the two materials at the interface. So, it can be controlled by surface treatment and change in the process.
Q6: When is rapid prototyping most appropriate?
Rapid prototyping is most appropriate for clinical trials and market validation stages. It is capable of making around 50-500 samples within 2-3 weeks. The cost of production is approximately 10%-20% of the mass production steel molds.
Q7: How to lower the unit price of medical parts?
By raising production volume, improving mold structure, shortening molding cycle, and reasonably selecting materials, you can achieve not only cost reduction but also efficiency improvement.
Q8: Where is your company (JS Precision) located?
JS Precision is a medical injection molding service provider located in China, who can offer ISO 13485 standard medical injection molding services from prototype to mass production stages.
Summary
At the heart of medical injection molding are compliance and precision, which are key elements for medical devices to go from the concept stage to large scale manufacturing.
With the expertise, tested solutions, and successful project examples, JS Precision can help you address three main problems: compliance auditing, precision control, and cost reduction.
If you want to get in touch with JS Precision's engineering team, send your 3D blueprints to avail a complimentary DFM evaluation and get a production ready plan from prototyping to industrialization.
Disclaimer
The contents of this page are for informational purposes only.JS Precision Services,there are no representations or warranties, express or implied, as to the accuracy, completeness or validity of the information. It should not be inferred that a third-party supplier or manufacturer will provide performance parameters, geometric tolerances, specific design characteristics, material quality and type or workmanship through the JS Precision Network. It's the buyer's responsibility Require parts quotation Identify specific requirements for these sections.Please contact us for more information.
JS Precision Team
JS Precision is an industry-leading company, focus on custom manufacturing solutions. We have over 20 years of experience with over 5,000 customers, and we focus on high precisionCNC machining,Sheet metal manufacturing,3D printing,Injection molding,Metal stamping,and other one-stop manufacturing services.
Our factory is equipped with over 100 state-of-the-art 5-axis machining centers, ISO 9001:2015 certified. We provide fast, efficient and high-quality manufacturing solutions to customers in more than 150 countries around the world. Whether it is small volume production or large-scale customization, we can meet your needs with the fastest delivery within 24 hours. Choose JS Precision this means selection efficiency, quality and professionalism.
To learn more, visit our website:www.cncprotolabs.com





